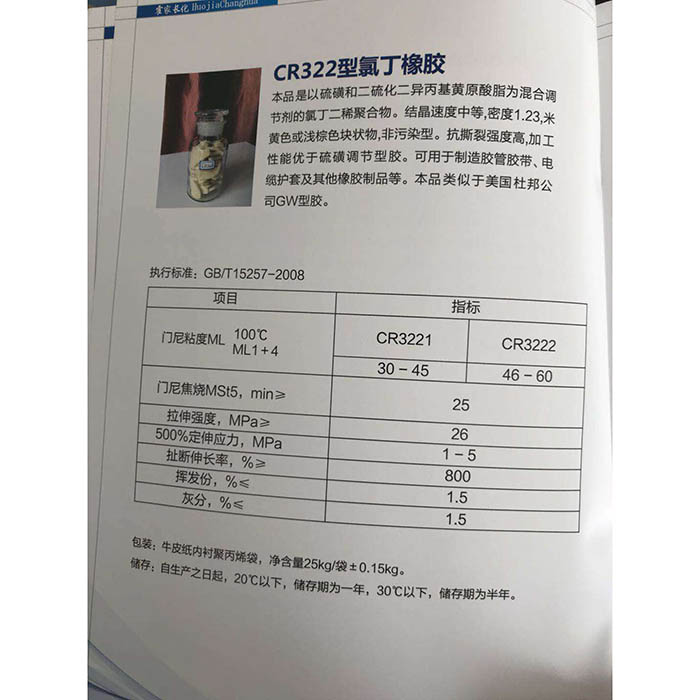
Sodium hydroxide
Sodium hydroxide, ane makemikoro emakemikari anonzi NaOH, anowanzozivikanwa se caustic soda, caustic soda uye caustic soda. Kana yanyungudutswa, inoburitsa kunhuhwirira kweammonia. Iro caustic yakasimbaalkali, iyo inowanzo mu flake kana granular fomu. Inonyungudika nyore mumvura (kana yakanyungudika mumvura, inopa kupisa) uye inoita alkaline solution. Pamusoro pezvo, ine deliquescent uye inotora nyore mhute yemvura (deliquescence) uye kabhoni dhaikisaidhi (kuora) mumhepo. NaOH ndeimwe yemakemikari anodiwa mumakemikari marabhoritari, uye zvakare imwe yeanowanzo makemikari. Iyo yakachena chigadzirwa haina ruvara uye yakajeka crystal. Density 2.130 g/cm. Kunyungudika 318.4 ℃. Nzvimbo yekubikira i1390 ℃. Zvigadzirwa zvemaindasitiri zvine zvishoma sodium chloride uye sodium carbonate, ari machena uye opaque makristasi. Iko kune blocky, flaky, granular uye tsvimbo-yakaita. Nyora huwandu 40.01
Sodium hydroxideinogona kushandiswa sealkaline yekuchenesa mumiririri mukurapa kwemvura, iyo inonyungudutswa mu ethanol uye glycerol; Insoluble mupropanol uye ether. Iyo zvakare inoshatisa kabhoni uye sodium pakupisa kwakanyanya. Disproportionation reaction nehalogen yakadai seklorini, bromine uye iodine. Neutralize neacids kugadzira munyu nemvura.
Zvenyama zvekupeta
Sodium hydroxide is white translucent crystalline solid. Mushonga wayo une mvura ine astringent kuravira uye sany kunzwa.
Kupeta deliquescence Inopeta mumhepo.
Kupeta kutorwa kwemvura
Solid alkali yakanyanya hygroscopic. Kana yaiswa kumhepo, inotora mamorekuru emvura mumhepo, uye pakupedzisira inonyungudika zvachose kuita mhinduro, asi mvura sodium hydroxide haina hygroscopicity.
Kupeta solubility
Kupeta alkalinity
Sodium hydroxide inozoparadzana zvachose kuita sodium ions uye hydroxide ions kana yanyungudutswa mumvura, saka ine huwandu hwealkali.
Inogona kuita acid-base neutralization reaction nechero protonic acid (inovawo yekaviri decomposition reaction):
NaOH + HCl = NaCl + H₂O
2NaOH + H₂SO₄=Na₂SO₄+2H₂O
NaOH + HNO₃=NaNO₃+H₂O
Saizvozvo, mhinduro yayo inogona kuita kaviri kuora kwekuita nemunyu mhinduro:
NaOH + NH₄Cl = NaCl +NH₃·H₂O
2NaOH + CuSO₄= Cu(OH)₂↓+ Na₂SO₄
2NaOH+MgCl₂= 2NaCl+Mg(OH)₂↓
Kupeta saponification reaction
Mune akawanda organic reactions, sodium hydroxide zvakare inobata basa rakafanana secatalyst, pakati peiyo inomiririra yakanyanya saponification:
RCOOR' + NaOH = RCOONA + R'OH
Kukoromora zvimwe
Chikonzero nei sodium hydroxide ichidzikira nyore kuita sodium carbonate (Na₂CO₃) mumhepo imhaka yekuti mweya une kabhoni dhaikisaidhi (co):
2NaOH + CO₂ = Na₂CO₃ + H₂O
Kana yakawandisa carbon dioxide ikaramba ichiunzwa, sodium bicarbonate (NaHCO₃), inowanzozivikanwa sesoda yekubheka, inogadzirwa, uye maitiro equation anotevera:
Na₂CO₃ + CO₂ + H₂O = 2NaHCO₃
Saizvozvo, sodium hydroxide inogona kuita neacidic oxides sesilicon dioxide (SiO₂) uye sulfur dioxide (SO):
2NaOH + SiO₂ = Na₂SiO₃ + H₂O
2 NaOH+SO (trace) = Na₂SO₃+H₂O
NaOH+SO₂ (yakanyanyisa) = NaHSO₃ (yakagadzirwa NASO uye mvura inopindirana neSO yakawandisa kugadzira nahSO)